Publication: Metabolically Primed Multipotent Hematopoietic Progenitors Fuel Innate Immunity
Publié dans: 2023
Auteurs: Jason Cosgrove, Anne-Marie Lyne, Ildefonso Rodriguez, Vincent Cabeli, Cecile Conrad, Sabrina Tenreira-Bento, Emilie Tubeuf, Erica Russo, Fanny Tabarin, Yannis Belloucif, Shayda Maleki-Toyserkani, Sophie Reed, Federica Monaco, Ann Ager, Camille Lobry, Philippe Bousso, Pablo Jose Fernández-Marcos, Hervé Isambert, Rafael Argüello, Leïla Perié
Résumé
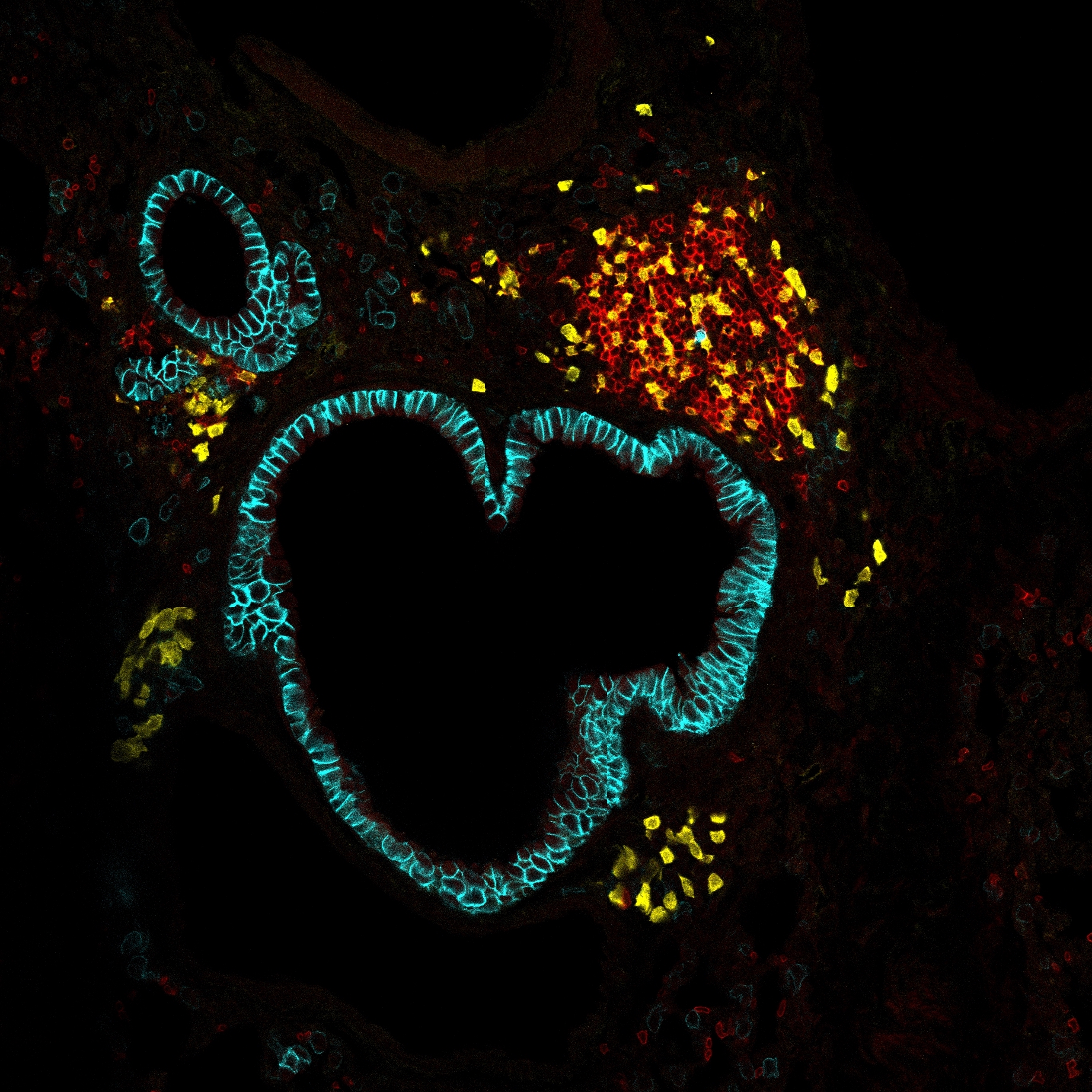
Following infection, hematopoietic stem and progenitor cells (HSPCs) support immunity by increasing the rate of innate immune cell production but the metabolic cues that guide this process are unknown. To address this question, we developed MetaFate, a method to trace the metabolic expression state and developmental fate of single cells in vivo . Using MetaFate we identified a gene expression program of metabolic enzymes and transporters that confers differences in myeloid differentiation potential in a subset of HSPCs that express CD62L. Using single-cell metabolic profiling, we confirmed that CD62L high myeloid-biased HSPCs have an increased dependency on oxidative phosphorylation and glucose metabolism. Importantly, metabolism actively regulates immune-cell production, with overexpression of the glucose-6-phosphate dehydrogenase enzyme of the pentose phosphate pathway skewing MPP output from B-lymphocytes towards the myeloid lineages, and expansion of CD62L high HSPCs occurring to support emergency myelopoiesis. Collectively, our data reveal the metabolic cues that instruct innate immune cell development, highlighting a key role for the pentose phosphate pathway. More broadly, our results show that HSPC metabolism can be manipulated to alter the cellular composition of the immune system.
Lien vers HAL – hal-04286084
Lien vers le DOI – 10.1101/2023.01.24.525166