Publication: Remodeling of skull bone channels regulates immune infiltration into the meninges during neuroinflammation.
Publié dans: Immunity 2026 Jan; 59(1): 116-128.e7
Auteurs: Eme-Scolan E, Gomes M, Bani A, Romano A, Kassem O, Roussel-Queval A, Casel B, Slimani L, Lawrence T, Rua R
Résumé
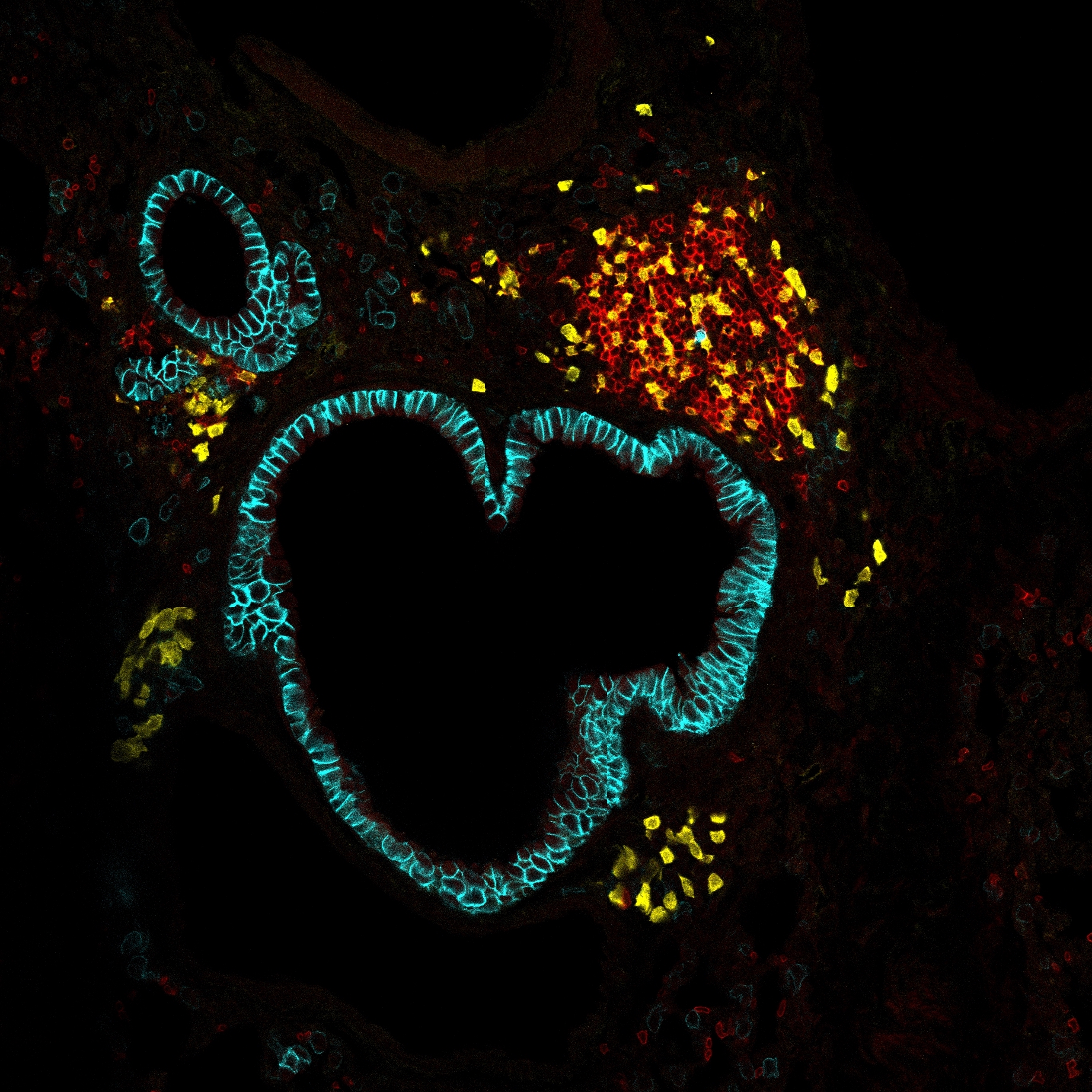
The meninges, located between the skull and brain, harbor immune cells that monitor the brain borders. Skull marrow communicates with the meninges via bone channels, enabling immune-cell trafficking, but little is known about bone channel formation and modulation. We found that bone channels were formed during the perinatal stage in mice, and we developed approaches to modulate them and assess their impact on meningeal immunity. Myeloid cell depletion with anti-colony-stimulating factor 1 receptor (αCSF1R) or targeted osteoclast inhibition with anti-receptor activator of nuclear factor kappa-B ligand (αRANKL) reduced channel size, whereas mechanoreceptor transient receptor potential vanilloid 4 (TRPV4)-driven bone remodeling enlarged them. Following channel manipulation, lymphocytic choriomeningitis virus (LCMV) infection showed reduced meningeal immune infiltration in αRANKL-treated mice and increased infiltration with TRPV4 activation. In an ex vivo skull assay, restricting channels impaired skull-derived immune-cell migration to the meninges, whereas enhancing remodeling promoted it. Our findings reveal that bone remodeling controls the skull-to-meninges axis and highlight its role in immune-cell migration into the meninges during neuroinflammation.
Lien vers Pubmed [PMID] – 41468886
Lien vers le DOI – 10.1016/j.immuni.2025.11.019